|
 |
Mechanism of Protein Unfolding
by Cellular Macromolecular Machines
|
 |
To be active most proteins must fold into well-defined three-dimensional structures. However, regulated unfolding is also critically important during the life cycle of many proteins, including proteins that are translocated across membranes and proteins that are degraded by ATP-dependent proteases. |
|
Some of these unfolding processes show surprising similarities. For instance, many proteins are targeted for unfolding by N- or C-terminal signal sequences, and often translational movement of the substrate protein occurs with unfolding. Despite the obvious biological relevance, and in contrast to protein folding, protein unfolding in vivo has not been studied.
Our laboratory studies the mechanism by which proteins are unfolded by translocases and proteases. |
|
|
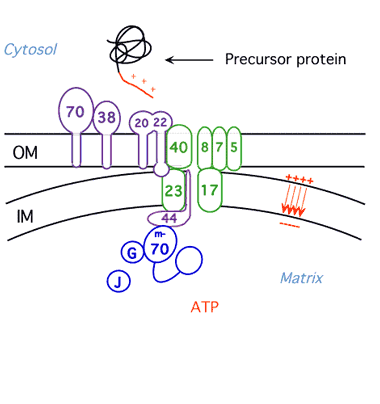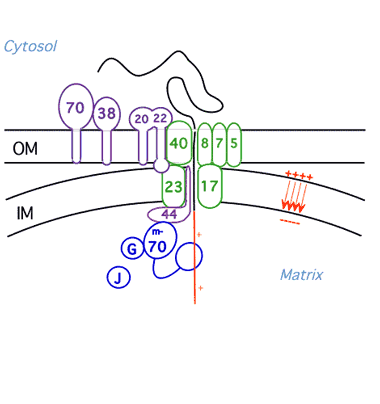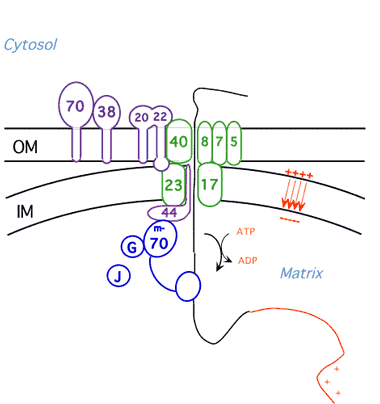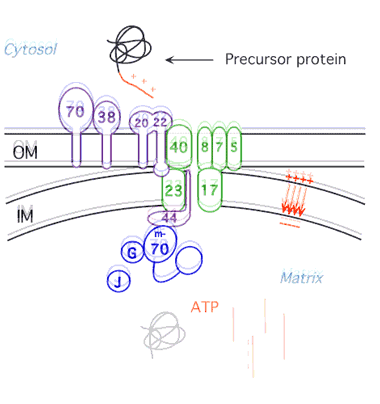
Protein Unfolding During Import into Mitochondria
|
We have shown that mitochondria and proteases unfold proteins by unraveling them from their targeting signals. We are now pursuing three lines of investigation: 1) identification of the components of the mitochondrial import machinery that unravel the protein during import employing methods of yeast cell biology and biochemistry; 2)characterization of the import motor using biophysical techniques; 3) investigation of the influence of three dimensional structure of proteins on their ability to be unfolded by the ATP-dependent proteases and the mechanism of partial degradation of transcription factors by the proteasome.
|
2003 Eli Lilly Award in Biological Chemistry
| [Home] | - | [Research Interest] | - | [Publications] | - | [Lab Members] | - | [Links] | - | [Contact Us] |
Matouschek Laboratory
Department of Molecular Biosciences